Experts Matter. Find Yours.
Connect for media, speaking, professional opportunities & more.
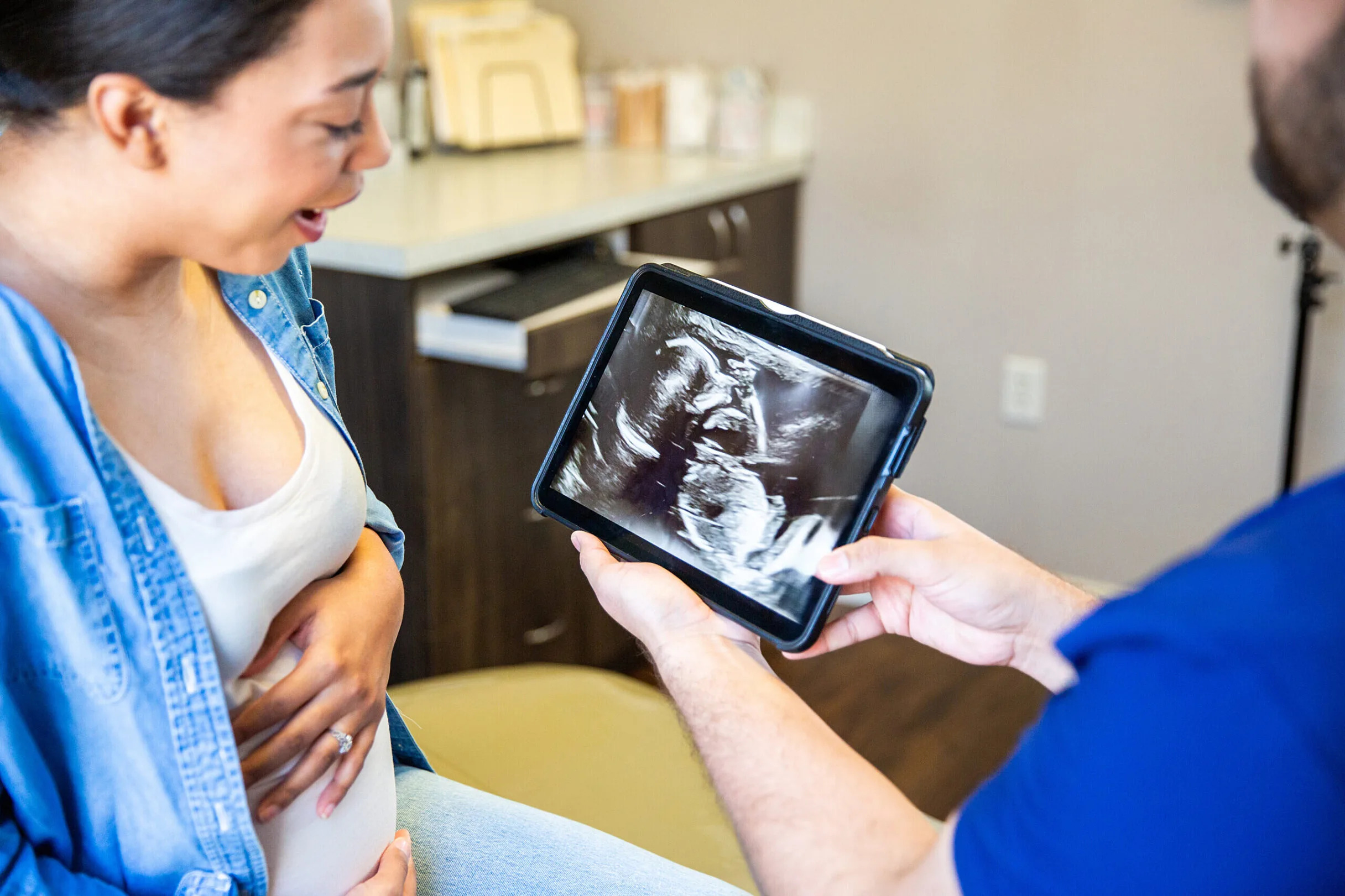
Study: Many pregnant women uncertain of marijuana risks even as use increases
Many pregnant women are unsure if it’s safe to use marijuana or products containing cannabidiol, an active compound in marijuana, even as they increasingly turn to them to combat morning sickness, anxiety or insomnia, a recent University of Florida Health study shows. The American College of Obstetricians and Gynecologists advises against the use of marijuana and cannabidiol, or CBD, during pregnancy. Marijuana use has been associated in some studies with adverse fetal neurodevelopmental outcomes. Evidence of cannabidiol safety is sparse in human studies, but researchers remain concerned that it might nonetheless pose a danger. CBD is not intoxicating. The UF Health researchers said their study shows a need for the medical community to better educate women about the potential hazards to the fetus from using marijuana, also called cannabis. One worry is that some people believe the spreading legalization of marijuana or CBD around the nation equates to the government giving its stamp of approval that the products are safe, researchers said. Medical marijuana is legal in Florida, although its recreational use is not. “If a medication is legal, we assume that maybe it’s safe, although other things like tobacco and alcohol are also legal and we know that those can be harmful to pregnancies,” said Kay Roussos-Ross, M.D., the study’s senior author and a professor in the UF College of Medicine’s Department of Obstetrics and Gynecology. “We see a good deal of data out there that shows that there is increased risk of psychiatric and behavioral issues related to marijuana use in pregnancy, but we need more,” she added. “We need more so that we can be correct in our assessments and our educational efforts to women of reproductive age who are using marijuana.” It’s difficult to quantify the rise of marijuana and CBD use during pregnancy, with most estimates showing an increase predating COVID-19. A 2021 federal survey reported 7.2% of pregnant women used marijuana. The UF Health study noted that emerging evidence from obstetrics care shows more pregnant women are trying the products, perhaps because of increased legalization. The study, published in Medical Cannabis and Cannabinoids, surveyed 261 women and used focus groups to explore participants’ perceptions of the products. The women were either pregnant, breastfeeding or caring for a child 5 years old or younger, and reported use of marijuana or CBD products, such as vapes, smoking, tincture oils or ointments. “There seems to be a disconnect,” said Amie Goodin, Ph.D., an assistant professor in the UF College of Pharmacy’s Department of Pharmaceutical Outcomes and Policy and the study’s lead author. “About one in six pregnant women are telling us, ‘Yes, I have used marijuana or a CBD product while I’m pregnant.’ But half are saying, ‘I don’t know what the risks are.’” About 40% of the pregnant women surveyed said they were unsure how risky it was to use marijuana once or twice a week during pregnancy, compared with 34.5% of women who were not pregnant when surveyed for the study but who had children. Asked the same question about CBD, more than 52% of pregnant women were unsure of the risk, compared with 41.8% for mothers who weren’t pregnant when surveyed. About 36% of pregnant women reported using marijuana, compared with 65% of mothers not currently pregnant, perhaps reflecting at least some increased caution among those in the former group. CBD use was 19.9% for pregnant women and 38.2% for women who were not pregnant. “Some women did mention that the legalization of marijuana has made marijuana more socially acceptable,” said study co-author Deepthi Varma, Ph.D., an assistant professor in the College of Public Health and Health Professions’ Department of Epidemiology. The researchers said they were especially concerned that women were even less sure of the safety of CBD use because it is widely available and often seen as harmless. “You might notice that it’s even something that you can buy at a gas station or a grocery store,” Goodin said. “CBD in a purified form has actually got an FDA approval to treat certain types of pediatric epilepsy on its own … but pharmaceutical-grade CBD is not quite the same thing as you would expect to get if you were purchasing CBD oil at a smoke shop or a gas station.”

Surprising finding could pave way for universal cancer vaccine
An experimental mRNA vaccine boosted the tumor-fighting effects of immunotherapy in a mouse-model study, bringing researchers one step closer to their goal of developing a universal vaccine to “wake up” the immune system against cancer. Published today in Nature Biomedical Engineering, the University of Florida study showed that like a one-two punch, pairing the test vaccine with common anticancer drugs called immune checkpoint inhibitors triggered a strong antitumor response in laboratory mice. A surprising element, researchers said, was that they achieved the promising results not by attacking a specific target protein expressed in the tumor, but by simply revving up the immune system — spurring it to respond as if fighting a virus. They did this by stimulating the expression of a protein called PD-L1 inside of tumors, making them more receptive to treatment. The research was supported by multiple federal agencies and foundations, including the National Institutes of Health. Senior author Elias Sayour, M.D., Ph.D., a UF Health pediatric oncologist and the Stop Children's Cancer/Bonnie R. Freeman Professor for Pediatric Oncology Research, said the results reveal a potential future treatment path — an alternative to surgery, radiation and chemotherapy — with broad implications for battling many types of treatment-resistant tumors. “This paper describes a very unexpected and exciting observation: that even a vaccine not specific to any particular tumor or virus — so long as it is an mRNA vaccine — could lead to tumor-specific effects,” said Sayour, principal investigator at the RNA Engineering Laboratory within UF’s Preston A. Wells Jr. Center for Brain Tumor Therapy. “This finding is a proof of concept that these vaccines potentially could be commercialized as universal cancer vaccines to sensitize the immune system against a patient’s individual tumor,” said Sayour, a McKnight Brain Institute investigator and co-leader of a program in immuno-oncology and microbiome research. Until now, there have been two main ideas in cancer-vaccine development: To find a specific target expressed in many people with cancer, or to tailor a vaccine that is specific to targets expressed within a patient's own cancer. “This study suggests a third emerging paradigm,” said Duane Mitchell, M.D., Ph.D., a co-author of the paper. “What we found is by using a vaccine designed not to target cancer specifically but rather to stimulate a strong immunologic response, we could elicit a very strong anticancer reaction. And so this has significant potential to be broadly used across cancer patients — even possibly leading us to an off-the-shelf cancer vaccine.” For more than eight years, Sayour has pioneered high-tech anticancer vaccines by combining lipid nanoparticles and mRNA. Short for messenger RNA, mRNA is found inside every cell — including tumor cells — and serves as a blueprint for protein production. This new study builds upon a breakthrough last year by Sayour’s lab: In a first-ever human clinical trial, an mRNA vaccine quickly reprogrammed the immune system to attack glioblastoma, an aggressive brain tumor with a dismal prognosis. Among the most impressive findings in the four-patient trial was how quickly the new method — which used a “specific” or personalized vaccine made using a patient’s own tumor cells — spurred a vigorous immune-system response to reject the tumor. In the latest study, Sayour’s research team adapted their technology to test a “generalized” mRNA vaccine — meaning it was not aimed at a specific virus or mutated cells of cancer but engineered simply to prompt a strong immune system response. The mRNA formulation was made similarly to the COVID-19 vaccines, rooted in similar technology, but wasn’t aimed directly at the well-known spike protein of COVID. In mouse models of melanoma, the team saw promising results in normally treatment-resistant tumors when combining the mRNA formulation with a common immunotherapy drug called a PD-1 inhibitor, a type of monoclonal antibody that attempts to “educate” the immune system that a tumor is foreign, said Sayour, a professor in UF’s Lillian S. Wells Department of Neurosurgery and the Department of Pediatrics in the UF College of Medicine. Taking the research a step further, in mouse models of skin, bone and brain cancers, the investigators found beneficial effects when testing a different mRNA formulation as a solo treatment. In some models, the tumors were eliminated entirely. Sayour and colleagues observed that using an mRNA vaccine to activate immune responses seemingly unrelated to cancer could prompt T cells that weren’t working before to actually multiply and kill the cancer if the response spurred by the vaccine is strong enough. Taken together, the study’s implications are striking, said Mitchell, who directs the UF Clinical and Translational Science Institute and co-directs UF’s Preston A. Wells Jr. Center for Brain Tumor Therapy. “It could potentially be a universal way of waking up a patient’s own immune response to cancer,” Mitchell said. “And that would be profound if generalizable to human studies.” The results, he said, show potential for a universal cancer vaccine that could activate the immune system and prime it to work in tandem with checkpoint inhibitor drugs to seize upon cancer — or in some cases, even work on its own to kill cancer. Now, the research team is working to improve current formulations and move to human clinical trials as rapidly as possible. While the experimental mRNA vaccine at this point is in early preclinical testing — in mice not humans — information about available nonrelated human clinical trials at UF Health can be viewed here.

AU scientists advance understanding of Nobel-winning immunology research
The 2025 Nobel Prize in Physiology or Medicine recognized the discovery of regulatory T cells, which are immune cells that maintain tolerance and prevent autoimmunity. At Augusta University, scientists have built upon that foundational work, uncovering how these cells function, fail and evolve across diseases like cancer and atherosclerosis. Regulatory T cells, or Tregs, are essential for controlling immune responses and preventing the body from attacking its own tissues. Early discoveries by Shimon Sakaguchi, MD, PhD, who identified the CD25 marker and later the transcription factor FoxP3, revealed how Tregs suppress immune activation. Sakaguchi, a distinguished professor at Osaka University in Japan, shared the Nobel Prize with Mary E. Brunkow, PhD, Princeton University, and Fred Ramsdell, PhD, University of California, Los Angeles. Sakaguchi once shared lab space with Nicholas Gascoigne, PhD, now a professor at AU’s Immunology Center of Georgia, where he studies T-cell signaling and differentiation — a connection that ties the global history of Treg research to the university’s ongoing expertise. Gascoigne’s research continues to illuminate how Tregs differentiate and signal, critical steps in ensuring immune tolerance to self. “Drs. Sakaguchi, Brunkow and Ramsdell have made enormous contributions to our understanding of how immunological tolerance works, so this prize is very well deserved,” Gascoigne said. “I was happy I could help Drs. Shimon and Noriko Sakaguchi when they needed lab space back in the early ‘90s. They were wonderful to work with.” At Augusta University, David Munn, MD, co-director of the Pediatric Immunotherapy Program of the Medical College of Georgia at Augusta University, made seminal discoveries showing that Tregs are not always stable. Munn’s team demonstrated that these cells could lose their suppressive identity and become “exTregs,” adopting pro-inflammatory characteristics under certain conditions. Munn also uncovered an entirely distinct tolerance mechanism through indoleamine 2,3-dioxygenase, a pathway independent of FoxP3 that regulates immune balance through tryptophan metabolism. “In particular, the work of Shimon Sakaguchi and Fred Ramsdell was profoundly influential on our early work, as we were just starting out to explore how the immune system is regulated in pediatric cancers,” Munn said. “The work of these Nobel laureates helped transform the scientific understanding of how the immune system responds — or fails to respond — in the setting of tumors.” Catherine “Lynn” Hedrick, PhD, co-director of the Immunology Center of Georgia, further expanded this understanding by showing that Tregs can convert into follicular helper T cells, offering new insight into how immune regulation can shift toward antibody production in chronic inflammatory diseases such as atherosclerosis. “Understanding how regulatory T cells can shift identities helps explain why the immune system sometimes loses balance in chronic disease,” Hedrick said. “By tracing how Tregs convert into other helper cells, we’re uncovering new therapeutic targets to restore immune harmony in conditions like atherosclerosis.” Building on these advances, Klaus Ley, MD, co-director of IMMCG, and his team have investigated how exTregs contribute to cardiovascular inflammation, identifying them in human tissues and clarifying their role in atherosclerosis. His lab also recently discovered human exTregs, providing the first direct evidence of these cells in people. Two postdoctoral fellows in Ley’s lab, Qingkang Lyu, PhD, and Smriti Parashar, PhD, continue this work, exploring how regulatory and ex-regulatory T cells influence chronic disease progression. Additionally, Dimitrios Moskofidis, MD, PhD, a professor based in the Georgia Cancer Center, contributed key early insights into immune tolerance and memory, defining how effector T cells persist or are deleted following infection. “Together, these discoveries place Augusta University and the Immunology Center of Georgia at the forefront of modern immunology, connecting molecular mechanisms of tolerance to real-world diseases and therapies,” Ley said. To connect with any of the experts or researchers in this article - simply contact AU's External Communications Team mediarelations@augusta.edu to arrange an interview today.
Heart valve developed at UC Irvine shines in early-stage preclinical testing
UC Irvine researchers designed and developed a minimally invasive replacement pulmonary heart valve. Created for pediatric patients, the device can be expanded as children grow, eliminating the need for multiple surgeries. The team successfully conducted laboratory and early-stage animal feasibility testing of the implant, crucial steps toward approval for human use. Irvine, Calif., June 23, 2025 — Researchers at the University of California, Irvine have successfully performed preclinical laboratory testing of a replacement heart valve intended for toddlers and young children with congenital cardiac defects, a key step toward obtaining approval for human use. The results of their study were published recently in the Journal of the American Heart Association. The management of patients with congenital heart disease who require surgical pulmonary valve replacement typically occurs between the ages of 2 and 10. To be eligible for a minimally invasive transcatheter pulmonary valve procedure, patients currently must weigh at least 45 pounds. For children to receive minimally invasive treatment, they must be large enough so that their veins can accommodate the size of a crimped replacement valve. The Iris Valve designed and developed by the UC Irvine team can be implanted in children weighing as little as 17 to 22 pounds and gradually expanded to an adult diameter as they grow. Research and development of the Iris Valve has been supported by the Eunice Kennedy Shriver National Institute of Child Health and Human Development; the National Heart, Lung, and Blood Institute; and the National Science Foundation. This funding has enabled benchtop fracture testing, which demonstrated the valve’s ability to be crimped down to a 3-millimeter diameter for transcatheter delivery and subsequently enlarged to 20 millimeters without damage, as well as six-month animal studies that confirmed successful device integration within the pulmonary valve annulus, showing valve integrity and a favorable tissue response. “We are pleased to see the Iris Valve performing as we expected in laboratory bench tests and as implants in Yucatan mini pigs, a crucial measure of the device’s feasibility,” said lead author Arash Kheradvar, UC Irvine professor of biomedical engineering. “This work represents the result of longstanding collaboration between our team at UC Irvine and Dr. Michael Recto at Children’s Hospital of Orange County built over several years of joint research and development.” Congenital heart defects affect about 1 percent of children born in the United States and Europe, with over 1 million cases in the U.S. alone. These conditions often necessitate surgical interventions early in life, with additional procedures required to address a leaky pulmonary valve and prevent right ventricular failure as children grow. The Iris Valve can be implanted via a minimally invasive catheter through the patient’s femoral vein. The Kheradvar group employed origami folding techniques to compress the device into a 12-French transcatheter system, reducing its diameter to no more than 3 millimeters. Over time, the valve can be balloon-expanded up to its full 20-millimeter diameter. This implantation method, along with the ability to begin treatment earlier in very young patients, helps mitigate the risk of complications from delayed care and reduces the need for multiple surgeries in this vulnerable population. “Once the Iris Valve comes to fruition, it will save hundreds of children at least one operation – if not two – throughout the course of their lives,” said Recto, an interventional pediatric cardiologist at CHOC who’s also a clinical professor of pediatrics at UC Irvine. “It will save them from having to undergo surgical pulmonary valve placement, as the Iris Valve is delivered via a small catheter in the vein and can be serially dilated to an adult diameter and also facilitate the future placement of larger transcatheter pulmonary valves – with sizes greater than 20 millimeters, like the Melody, Harmony and Sapien devices – if needed.” Kheradvar said that the next phase of preclinical testing of the Iris Valve is funded by the Brett Boyer Foundation, which is committed to supporting research into treatments for congenital heart disease. “We are actively engaged with the U.S. Food and Drug Administration to define and carry out the required experiments and documentation for first-in-human authorization of the Iris Valve,” Kheradvar said. “Our team is urgently advancing the Iris Valve through preclinical studies to enable its clearance for first-in-human use. This is a critical step toward providing toddlers – who currently have no viable minimally invasive treatment until they reach the 45-pound threshold – with a much-needed option.” First co-author Nnaoma Agwu, a biomedical engineering Ph.D. candidate at UC Irvine, said: “The development of the Iris Valve required a strong and knowledgeable team that understood the clinical and mechanical design requirements. This accomplishment would not have been possible without the collaboration of talented clinicians, veterinarians and engineers. With this milestone reached, we are rigorously advancing the Iris Valve’s development, setting our sights on human clinical trials.” Joining Kheradvar, Recto and Agwu as co-authors of the article in Journal of the American Heart Association were Daryl Chau, a recent UC Irvine master’s graduate; Gregory Kelley and Tanya Burney, both research specialists at UC Irvine, with Burney also affiliated with the Beckman Laser Institute; Ekaterina Perminov, a clinical veterinarian with UC Irvine’s University Laboratory Animal Resources; and Christopher Alcantara, a radiology technician at CHOC. About UC Irvine’s Brilliant Future campaign: Publicly launched on Oct. 4, 2019, the Brilliant Future campaign aims to raise awareness and support for the university. By engaging 75,000 alumni and garnering $2 billion in philanthropic investment, UC Irvine seeks to reach new heights of excellence in student success, health and wellness, research and more. The Samueli School of Engineering plays a vital role in the success of the campaign. Learn more by visiting https://brilliantfuture.UC Irvine.edu/the-henry-samueli-school-of-engineering About the University of California, Irvine: Founded in 1965, UC Irvine is a member of the prestigious Association of American Universities and is ranked among the nation’s top 10 public universities by U.S. News & World Report. The campus has produced five Nobel laureates and is known for its academic achievement, premier research, innovation and anteater mascot. Led by Chancellor Howard Gillman, UC Irvine has more than 36,000 students and offers 224 degree programs. It’s located in one of the world’s safest and most economically vibrant communities and is Orange County’s second-largest employer, contributing $7 billion annually to the local economy and $8 billion statewide. For more on UC Irvine, visit www.uci.edu. Media access: Radio programs/stations may, for a fee, use an on-campus studio with a Comrex IP audio codec to interview UC Irvine faculty and experts, subject to availability and university approval. For more UC Irvine news, visit news.uci.edu. Additional resources for journalists may be found at https://news.uci.edu/media-resources.

ChristianaCare and Children’s Hospital of Philadelphia (CHOP) today announce a new strategic affiliation to expand access to world-class pediatric care in Delaware and surrounding communities. The collaboration, which is expected to begin in spring 2026, will enhance access to specialized pediatric care by bringing CHOP’s renowned expertise closer to home for families in the communities ChristianaCare serves in Delaware, Maryland, Pennsylvania and New Jersey. By integrating CHOP’s services with ChristianaCare’s established network, the collaboration will improve care delivery, making advanced pediatric treatments more accessible and reducing the need for families to travel long distances for care. A Bold Step Forward in Pediatric Care “This collaboration brings together two amazing organizations that are already nationally recognized for clinical excellence—and we’re going to be even better together,” said Janice E. Nevin, M.D., MPH, president and CEO of ChristianaCare. “Our affiliation with CHOP marks a bold step forward in our mission to provide the highest quality care to children and families across the communities we serve. These are uncertain times for many parents with young children in need of routine or complex medical care. ChristianaCare and CHOP are joining forces to provide world-class care, close to home, at a time when it’s needed most.” “Bringing seamless, family-centered care to our patients is a priority at Children’s Hospital of Philadelphia,” said Madeline Bell, CEO at Children’s Hospital of Philadelphia. “As one of the region’s most dynamic health systems, ChristianaCare has earned a national reputation for its bold approaches to both clinical care and innovation. Our new affiliation is driven by a shared vision: redefining what is possible in health care and ensuring that every patient — at every stage of life — receives the highest standards of care, from infancy through adulthood.” “Our team at ChristianaCare is excited to work closely with Children’s Hospital of Philadelphia to bring even more specialized care to our community,” said Megan Mickley, M.D., chair of Pediatrics at ChristianaCare. “CHOP’s leadership in pediatric medicine will complement the outstanding care already provided at ChristianaCare, ensuring that our youngest patients receive the most advanced treatments in a compassionate and supportive environment.” Initial Focus and Future Growth This affiliation between ChristianaCare and CHOP represents a significant increase in access to world-class pediatric care for communities in Delaware, Maryland, southeast Pennsylvania and southern New Jersey. CHOP, a global leader in pediatric care and research for over 165 years, is consistently ranked among the top children’s hospitals in the country, known for pioneering breakthroughs across a breadth of pediatric care areas. The affiliation will include all ChristianaCare hospitals and locations where pediatric emergency and inpatients are cared for. The collaboration will initially focus on pediatric and neonatal services. Patients at ChristianaCare will have access to CHOP’s expertise in rare and complex diseases and a wide range of newborn and pediatric services, including surgery, cardiology, neurology, radiology, ophthalmology and genetics, as well as educational and research opportunities. As the collaboration grows, it will explore further opportunities to expand pediatric care and improve health care delivery in the region.

Maureen Leffler, D.O., Named ChristianaCare’s Chief Wellbeing Officer
Maureen “Mo” Leffler, D.O., MPH, has been appointed chief wellbeing officer of ChristianaCare, effective Nov. 25. In her role, Leffler leads the ChristianaCare Center for WorkLife Wellbeing and strategies to enhance the professional fulfillment and well-being of ChristianaCare’s nearly 14,000 caregivers, overseeing advocacy programs and initiatives to optimize their experience and foster a culture of well-being throughout the organization. She works closely with leaders across key departments to address factors impacting caregiver well-being. Leffler most recently served as the inaugural chief wellbeing officer at Nemours Children’s Health, where she helped the organization to achieve the 2022 Joy in Medicine distinction from the American Medical Association for prioritizing proven efforts to enhance the professional fulfillment of physicians. There, she established a Center for Associate Wellbeing; led the first systemwide assessment to strategically address well-being and burnout; and implemented a peer support program and expanded the scope of resources available to support the emotional and mental health needs of employees. In collaboration with organizational leaders, she supported targeted clinical team assessments and systems-based interventions to foster well-being. Prior to this role, Leffler served as a pediatric rheumatologist at Nemours and as an assistant professor of pediatrics in the Division of Rheumatology at Thomas Jefferson University. Since 2017, Leffler has served as the course director of the Chief Resident Leadership Training Program for the Accreditation Council for Graduate Medical Education (ACGME). In response to the COVID-19 outbreak, she co-chaired ACGME’s National Task Force on Well-Being. She and her team developed a national graduate medical education well-being community, which she continues to convene, to understand the evolving challenges and share strategies to improve well-being. She represents the ACGME as a coach for the National Academy of Medicine Action Collaborative on Clinical Well-Being and Resilience. She also serves as a consultant to the Professional Satisfaction team at the American Medical Association. Leffler earned her medical degree from the Philadelphia College of Osteopathic Medicine, followed by a residency in pediatrics at Nemours and Thomas Jefferson University Hospital, where she served as chief resident. She subsequently trained in pediatric rheumatology at Nemours and Jefferson. She also earned a Master of Public Health from Temple University and studied chemistry at St. Joseph’s University. Recently, Leffler completed the Georgetown Executive Leadership Certification Program.

ChristianaCare Honored for Emergency Nursing Excellence
ChristianaCare has earned the 2024 National Certification Champion Award for health systems from the Board of Certification for Emergency Nursing (BCEN), a leading authority in nursing specialty certification across the emergency care spectrum. ChristianaCare is the only national winner in its category. “ChristianaCare is honored to be named the BCEN National Certification Champion,” said Danielle Weber, DNP, MSM, RN-BC, NEA-BC, chief nurse executive at ChristianaCare. “This prestigious recognition speaks to the passion and dedication of our incredible nursing teams, guided by our values of love and excellence, who make the difference for patients during some of their worst moments, going above and beyond to deliver quality care.” “It’s a privilege to be a part of a team that is committed to nursing excellence, practicing at the top of their license through specialty certification,” Weber continued. “This award represents the culmination of years of focused attention by nursing leadership to promote and facilitate ED nurse certification coupled with a highly motivated and passionate nursing staff who are dedicated to their patients as well as their professional development. We are so proud of our certified nurses and their commitment to lifelong learning.” According to BCEN, nursing specialty certification independently validates a registered nurse’s advanced knowledge, clinical judgment and professionalism across an entire nursing specialty. Specialty board certification of nurses helps assure patients and their families that they are receiving the highest level of nursing care. A growing body of research links nursing specialty certification to improved patient care, safety and outcomes. “We congratulate ChristianaCare on its commitment to nursing excellence,” said BCEN CEO Janie Schumaker, MBA, BSN, RN, CEN, ICE-CCP, CENP, CPHQ, FABC. “The 2024 BCEN National Certification Champions show us how nursing specialty certification empowers nurses, elevates patient care and helps ensure communities of every size have access to advanced emergency and trauma care.” BCEN is an independent not-for-profit organization that offers nursing specialty certification programs for nurses across the emergency care spectrum. Over 50,000 registered nurses specializing in emergency, pediatric emergency, flight, critical care ground transport, trauma and burn nursing hold one or more BCEN certification.

Stephen Pearlman, M.D., Honored for Excellence in Neonatology Education
Stephen Pearlman, M.D., MSHQS, will receive the 2024 Avroy Fanaroff Neonatal Education Award at the American Academy of Pediatrics’ annual meeting in September. This honor, presented by the AAP’s section on Neonatal-Perinatal Medicine, recognizes an educator who makes outstanding contributions in neonatal-perinatal medicine for health care students, professionals or the public. Pearlman, an expert in neonatology, intensive care and pediatrics, is clinical effectiveness officer for acute care at ChristianaCare. He is also professor of pediatrics at Sidney Kimmel College of Medicine at Thomas Jefferson University. “Stephen is deeply committed to excellence in neonatal education and the highest quality care,” said Kert Anzilotti, M.D.,MBA, system chief medical officer at ChristianaCare. “As a faculty member, physician and leader, he continues to make a lasting impact at ChristianaCare and beyond with his pioneering initiatives in quality improvement and safety in health care.” A faculty member for almost 40 years, Pearlman has served ChristianaCare in clinical, educational and administrative roles. Among them, he was chair of the Pediatric and Neonatal Safety Committee, director of Neonatal Quality Improvement, associate director of Neonatology and director of Pediatric Medical Education. Pearlman has led initiatives that have been spotlighted by the federal government as exemplars of how to improve safety at health systems. He developed an innovative quality-improvement curriculum for neonatal fellows, which the Organization of Neonatal-Perinatal Training Program Directors adopted. “I’m passionate about educating clinicians about ways to improve the quality of patient care, so it’s humbling to receive this recognition,” Pearlman said. “I am honored to have been selected as a recipient of this award.” Pearlman is also an associate editor of Quality Improvement for the Journal of Perinatology and an executive committee member of the American Academy of Pediatrics’ Perinatal Section.

Innovative EEG Brain Monitoring Program Provides Optimal Care for Critically Ill Patients
ChristianaCare has launched an innovative electroencephalogram (EEG) brain monitoring program that represents a significant leap forward in the diagnosis and management of neurological conditions. The easy-to-use EEG program is the first of its kind that can be performed at the bedside to measure the electrical activity of the brain, providing a vital sign for brain function to help diagnose seizures more quickly. ChristianaCare is the first hospital system in Delaware to use the Ceribell point-of-care EEG monitoring system, which can reduce the time it takes to diagnose certain neurological conditions from hours to mere minutes. Using the system, clinicians have immediate access to EEG information so they can triage at-risk patients in just five minutes and monitor patients for treatment optimization. “With this new program, our team of expert clinicians will have the diagnostic information they need to provide high-risk patients with the right care at the right time, ensuring the best outcomes for our patients and their families,” said Kim Gannon, M.D., Ph.D., service line leader for Neurosciences at ChristianaCare. New technology detects ‘silent seizures’ Critically ill patients are at high risk of harmful brain electrical discharges called seizures. Some of these patients experience a type of “silent seizure” with no noticeable symptoms (non-convulsive) that can only be detected using EEG. If prolonged, non-convulsive seizures can lead to permanent brain injury and higher risk of morbidity and mortality. Demonstration of EEG device on patient at ChristianaCare Newark Campus. As a result, guidelines from the Neurocritical Care Society recommend EEG should be initiated within 15-60 minutes when these seizures are suspected. Meeting these guidelines has proven difficult due to the limitations of conventional EEG systems, which were not designed for use in emergency situations. Even top academic centers that have 24/7 EEG capabilities may experience wait times of four hours or more for conventional equipment. When relying on clinical judgement alone while waiting for these conventional EEG systems, diagnostic accuracy has been shown to be only slightly better than chance (65%). The value of this new technology for patients is that it provides accurate results quickly so that the care team can intervene early. “The neuroscience and critical care teams at ChristianaCare believe that ‘time is brain’ not only applies to stroke but also when dealing with seizure,” Gannon said. Gannon is referring to the fact that for every minute that passes when someone is having a stroke, 1.9 million brain cells are lost, increasing the chance of disability or death. That same kind of speed and urgency can now more easily be brought to bear for patients when a seizure is suspected. “This monitoring system is easy to use and can be set up in about five minutes,” said Richard Choi, D.O., medical director of the Neurocritical Care Unit at Christiana Hospital. “It consists of a simple headband, pocket-sized recorder with intuitive software and an on-line portal for remote viewing. Using the system, we can review EEG data, assess response to treatment and optimize care, all in real-time.” Neurosciences at ChristianaCare ChristianaCare’s multidisciplinary neurosciences team provides comprehensive and advanced care for neurologic illnesses across the acute and ambulatory settings. As the largest and most comprehensive neurology practice in Delaware with more than 55,000 patient visits last year, ambulatory subspecialties include stroke, epilepsy, multiple sclerosis, movement disorders, neuromuscular disorders, headaches/migraines, pediatric neurology and Botox specialists. The inpatient team of experts includes neurosurgeons, neurointerventional surgeons, neurocritical care physicians and vascular neurologists. The Newark Campus serves as the only comprehensive stroke center in the state and includes the only Epilepsy Monitoring Unit in Delaware.

New center helps meet growing need for pediatric care in our community Families in northern Delaware and surrounding areas have a new option for pediatric care. Today ChristianaCare opened a new 14-bed Pediatric Care Center that provides 24/7 combined short-stay inpatient and emergency care for children and teens. The new center is located on the first floor of the Center for Women’s & Children’s Health on ChristianaCare’s Newark Campus. “This new facility is an exciting step in our continued journey to create the absolute best care for children and families in our community,” said David Paul, M.D., chair of the Department of Pediatrics. “The Pediatric Care Center will significantly improve access to care for children, enhance the patient experience and address the needs of families who want high-quality care in a child-friendly environment.” The 24-hour Pediatric Care Center will be able to evaluate and manage 90% of the pediatric cases currently seen in the Christiana Hospital Emergency Department. The center expects to care for an estimated 6,300 patients the first year, with volume projected to grow 5% each year. The Pediatric Care Center also provides expert care to children and adolescents with behavioral health emergency needs and appropriate referral sources for follow-up care. “Our new Pediatric Care Center makes it easier than ever for families and children to receive excellent care when they need it, in a special space designed just for them,” said Sharon Kurfuerst, Ed.D., OTR/L, FACHE, system chief operating officer at ChristianaCare and President, Union Hospital. “The center will care for the special needs of pediatric patients, making it convenient for them to receive dedicated, expert resources for hospital-based, non-trauma emergency and inpatient care all in one location.” The 8,400 square-foot facility provides 14 beds for emergency and short-stay inpatient and observation care. “We offer 24/7 pediatric emergency care 365 days a year,” said Megan Mickley, M.D., MBA, FAAP, FACEP, medical director, who is board certified in Pediatrics and Pediatric Emergency Medicine and fellowship-trained in Pediatric Emergency Medicine and Emergency Ultrasound. “The Pediatric Care Center team is a diverse, multidisciplinary group led by pediatric physicians trained in a variety of backgrounds, including emergency medicine and hospital medicine,” she said. “Our goals are not only to provide exceptional quality of care for children, but also to improve access to and expand pediatric care services for our neighbors. We look forward to being a trusted partner for pediatric care in our community.” About the Center for Women’s & Children’s Health The Center for Women’s & Children’s Health, which opened in a state-of-the-art facility in 2020 on ChristianaCare’s Newark campus, represents a new standard of care for our community. It is the region’s only National Community Center of Excellence in Women’s Health and offers innovative, patient-centered care for mothers, babies and families. The center provides private rooms for mothers and families after delivery and is one of the only hospitals in the United States to provide couplet care in the Neonatal Intensive Care Unit, keeping mother and baby together even if they both require medical care.




